
- Application Services
- Next-Gen App Development
- Microsoft services
-
-
- Microsoft Fabric Consulting ServicesBuild advanced data pipelines to optimize Microsoft Fabric environment
- Microsoft Fabric Implementation ServiceGet powerful insights and simplify your data operations
- Microsoft BI SolutionsOrganize & analyze data for strategic advantage
- Real Time Intelligence Microsoft FabricTransform high-velocity data into instant, actionable insights
- Power BI Consulting ServicesCentralize, analyze, & visualize data for smarter decisions
-
- Microsoft Fabric Migration ServiceUnifying data for powerful analytics and AI-driven growth
- Microsoft Fabric Data Engineering ServicesEnd-to-end data engineering for large-scale data processing
- Microsoft Fabric Data ScienceExtract deep business insights and anticipate future trends
- Fabric Managed Services & SupportAchieve seamless operations, peak performance, and accelerated insights
-
-
- Mobile Application Development
- Modernization
-
-
- Integration ServicesGet seamless integration of various 3rd party services or existing systems for smooth operations of your mobile apps
- Cross PlatformsExcellent user experience and functionality of native apps with multi-platform compatibility
- Legacy System Modernization ServicesLegacy system modernization empowers organizations so that they can work more efficiently and intelligently.
-
-
- DevOps
- Software Testing
-
-
- AI Enabled TestingLeverage AI for testing the quality of your software products
- Functional TestingAutomated Functional Testing for improved quality & faster go-to-market
- STQC CertificationQuality, Excellence, and Innovation – The Hallmarks of Suma Soft
- Performance TestingEliminate performance bottlenecks of business-critical apps to run under expected, peak loads
-
- Scriptless Automation SolutionsFast, reliable, and easy testing with our “Ready-to-Deploy” automation framework
- Devops TestingOptimize testing in CI/CD pipelines for greater agility, efficiency, and quality
- AA CertificationEmpaneled Certifier for Account Aggregator (AA) Ecosystem
- ABDM Ecosystem Sandbox Exit ProcessWe Certify Your API for ABDM Sandbox Integration And Exit Process
-
-
- AI/ML Services
- AI/ML ServicesStay ahead of the curve with our advanced AI/ML capabilities
- Data Engineering ServicesDrive data-driven decision-making with our agile and customizable data engineering solutions
- Software Product Engineering ServicesTransform your concepts into market-leading products with our strategic product engineering approach
- VaniePartner with Us for Innovative Web App Solutions!
- Digital Public Infrastructure
- Account AggregatorGet certified using Daksh DEPA Validator to go live on the AA network.
- Ayushman Bharat Digital Mission (ABDM)Get certified using Daksh DEPA Validator to go live on the ABDM network.
- Beckn Protocol ImplementationImplement Beckn protocol to go live on India Stack network.
- ONDC CertificationGet certified using Daksh DEPA Validator to go live on the ONDC network.
- O Rickshaw Pune’s First ONDC Mobility App.
- Verifiable Credentials (VC)Securely issue digital credentials for trusted identity and data exchange.
- Unified QR CodeA single QR code to securely access and share information.
- e-KYC (Electronic Know Your Customer)Digitize and automate identity verification for customer onboarding.
- Data ExchangeSecure, consent-based data sharing between organizations and individuals.
- Consent ManagerEmpowering organizations to control data consent preferences.
- Business Services
- Business Process Management
-
-
- Healthcare BPO ServicesRound the clock coverage for most critical industry
- Logistics BPO & Supply Chain ManagementSmarter supply chains Built for today and ready for tomorrow
- Data Entry ServicesEnsure the accuracy and security of your data with Suma Soft
- Underwriting support servicesStreamline underwriting processes , increase accuracy, reduce risk with expert support services.
- Finance and Accounting ManagementImproving Account Payable and Receivables Through Artificial Intelligence
-
- Logistics BPO ServiceOptimize logistics with efficient operations, reduced costs, and our improved visibility.
- Customer Support ServicesSmart use of technology makes all the difference in CX
- REIT Management ServicesProductivity for You. Transparency for Your Investor
- OCR Outsourcing ServicesAchieve faster, accurate and reliable data capture with OCR outsourcing solutions.
- Claims ManagementState-of-the-art technology to assist in responding to rapidly-evolving claim situations
-
-
- Tech Support
-
-
- Desktop SupportResponsive, Day-to-Day Tech Assistance
- Network SupportCreate a comprehensive network security strategy for protecting the enterprise end-to-end
- Active Directory SupportMinimize the risk of disrupting critical business apps while optimizing the performance of MS infrastructure
- Microsoft Exchange SupportStay on top of imminent threats to achieve a glitch-free network with rapid Microsoft Exchange support
-
- L1 & L2 SupportOptimize performance with round-the-clock technical support cater to your business goals
- Remote MonitoringKeep your IT systems running smoothly and ensure the stability and security of your IT environment for the workforce
- NOCGet unrivaled NOC services & scale immediately to enhance your bottom-line profitability with 24*7 services
-
-
- Hyperautomation
-
- Cyber Security
-
-
- AssessmentTest your applications and networks to identify & fix vulnerabilities exposing your sensitive assets
- Cloud SecurityIntelligence and expertise providing a new level of cyber-immunity
- Managed SecurityComprehensive security architecture with flexible enforcement for advanced protection
- Digital ForensicsEmpower your organization to access, manage and leverage Digital Data
-
-
- ServiceNow
- ServiceNow Change and Release Management ServicesStreamline change and release for faster, reliable deployments.
- ServiceNow Agentic AI ServicesAutomate complex workflows and enhance decision-making with AI.
- ServiceNow Development ServicesBuild custom applications to enhance your ServiceNow platform.
- ServiceNow Managed ServicesOngoing ServiceNow support and optimization for peak performance.
- ServiceNow Integration ServicesOngoing ServiceNow Integration Services
- ServiceNow Implementation ServicesExpert ServiceNow implementation for customizable business solutions.
- ServiceNow Migration ServiceAchieve seamless platform transitions with smooth ServiceNow migration.
- ServiceNow Consulting ServicesStrategic ServiceNow consulting for optimal platform utilization.
- ServiceNow ITBM Implementation ServicesImplement ITBM for strategic IT planning and execution.
- ServiceNow ITSM Implementation ServicesPlan and streamline IT service management with expert implementation services.
- ServiceNow Chatbot DevelopmentEnhance user experience with intelligent ServiceNow chatbot solutions.
- ServiceNow IT Operations Management ServicesOptimize Your IT Operations with automated workflows and efficient service delivery.
- ServiceNow CSM ImplementationImprove customer service with efficient ServiceNow CSM implementation.
- ServiceNow GRC Implementation ServicesManage governance, risk, and compliance with ServiceNow GRC.
- ServiceNow Asset Management ServicesControl IT assets, reduce costs, and ensure compliance with ServiceNow Asset Management.
- ServiceNow Security Operations ServicesStrengthen security posture with ServiceNow security operations management.
- ServiceNow HR Service Delivery ServicesEnhance employee experiences with automated HR service management.
- ServiceNow Financial Management ServicesStreamline financial operations and improve decision-making with ServiceNow.
- ServiceNow Project Portfolio Management ServicesPrioritize projects and manage resources for successful delivery.
- ServiceNow Resource Management ServicesStreamline resource allocation for project efficiency and success with ServiceNow Resource Management.
- Business Process Management
- Digital
- Cloud
-
-
- Cloud App Development ServicesGet high security, efficient performance, and excellent operations with cloud apps
- Cloud Testing ServicesDelivering highly polished solutions to help you reduce costs and meet deadlines
- Cloud Migration ServicesA secure cloud single sign-on solution that IT, security, and users will surely love
-
- Enterprise Mobility
- CRM
- Microsoft Dynamics 365 ServicesStreamline business processes with superior BI, automation, and key customer insights
- Custom CRM Development ServicesDigitize processes, enhance everyday managerial activities and engage more customers
- Salesforce Consulting ServicesTransform Your Business with Expert Salesforce Guidance – Elevate Performance and Drive Growth!
- Salesforce testing servicesAchieve end-to-end automation for Salesforce testing
- E-Learning
- Collaboration
- Dusmile Platform
- Cloud
- Insights
- Industries
- Industry We Serve
- Banking And FinanceAccelerate your journey with fintech innovation to reshape customer experience
- GovernmentFully-managed environment for Government, Built for security and compliance
- E-CommerceBest-of-breed technologies to enhance CX and increase revenue to maximize business needs
- E-LearningReimaging education – from remote to hybrid learning
- Logistics & DistributionChanging the landscape of supply chain management
- AutomotiveRevolutionizing automotive development for the digital future
- HealthcareDelivering better experiences, better insights, and better care for better health
- ManufacturingLeverage our deep expertise to modernize your manufacturing and industrial technologies and processes.
- Industry We Serve
- About Us
- About Suma Soft
- HistoryDecades of experience and expertise matched with dedicated professionals.
- LeadershipA leadership that drives Suma Soft to meet its overall goals.
- CertificationsOur recognition as a trusted technology company for orchestrating a successful business transformation
- Our PartnersBringing together the best technologies and practices to offer a prosperous digital future
- Annual ReturnSuma Soft’s Yearly Financial Annual Return Report
- About Suma Soft
- Careers
- Contact Us
Clinical trials are an important part of any clinical research. They are used to evaluate the safety and efficacy of new treatments, drugs, and medical devices. Without accurate budgeting for these trials, the entire research process can become a costly and lengthy endeavor.
The cost of conducting a clinical trial can vary widely depending on the number of participants, the type of research being conducted, and the amount of data being collected. Budgeting for these trials must consider all these factors and more. It is important to have a clear and accurate budget of clinical trials prepared to ensure that the projected revenue of the hospital is accurate.
It is important to have a budget that covers all the expenses associated with the trial, including prescreening and screening of patient, participant compensation, laboratory testing, and equipment costs.
Periodic review and timely revision of the budget is important in case of amendments to clinical trial contract. A Quality Assurance procedure for review of budget helps to ensure that all the latest contractual terms are correctly translated into budget and discrepancies can be fixed on timely manner.
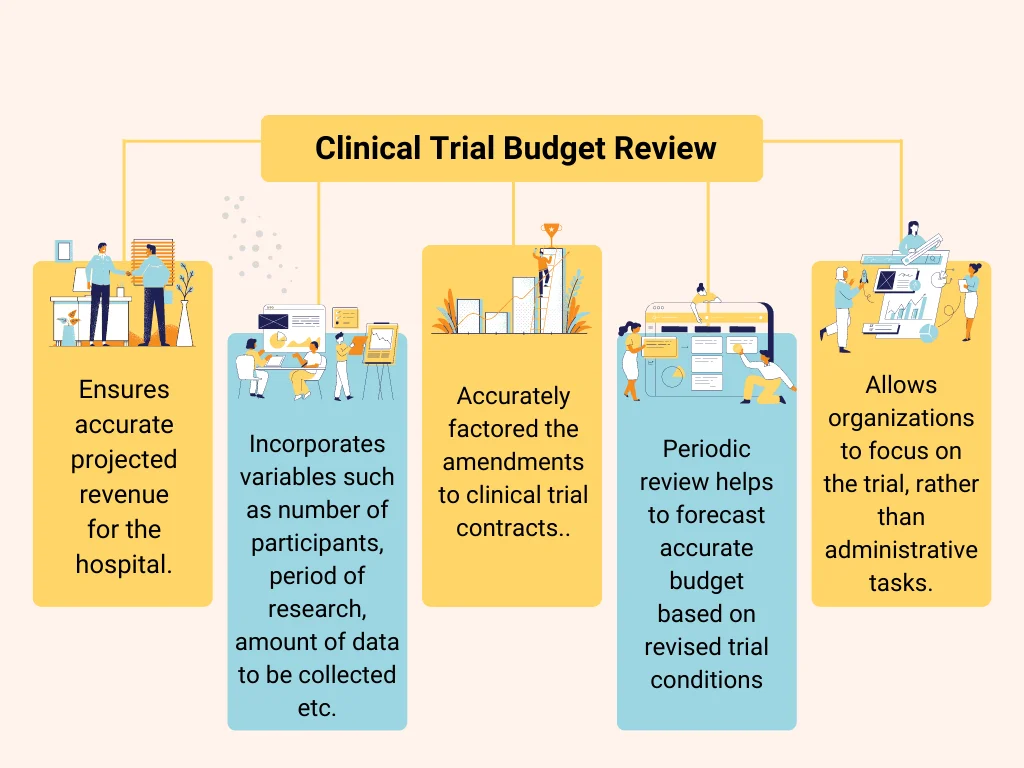
How Suma Soft is helping healthcare industries to prepare and review accurate budget for clinical trials?
Suma Soft provides a range of valuable services to help healthcare organizations manage and budget for clinical trials.
Our team of analysts have experience of preparation of budgets, corrections to it based on any amendments and audit of budget based on contractual terms. It helps the healthcare organizations/hospitals to save lot of time and money, allowing them to focus on the actual trial rather than worrying about administrative tasks.
Recent Posts
- Top 10 ITSM Consulting Companies in India for 2026 April 20, 2026
- DevOps in Software Product Engineering: Streamlining Development and Operations September 10, 2024
- Accelerate Your Business with Our Data Engineering Solutions: A Case Study August 20, 2024
- The Role of Cloud Computing in Modern Data Engineering Solutions June 25, 2024
- Why 70% of Enterprises Are Choosing Hybrid Apps and How They Benefit May 24, 2024
- How Building a Native App Can Increase Customer Engagement by 3X May 20, 2024
- What Competitive Advantages Do PWAs Offer Over Traditional Mobile Apps? May 16, 2024
- 5 Must-Have Features for Enterprise-Level iOS Apps May 14, 2024
- 5 Ways to Supercharge Your Android App with AI and Machine Learning May 8, 2024
- Comparing Traditional ETL vs. Modern Data Engineering Approaches April 24, 2024
